The percentage by volume of C3H8 in gaseous mixture of C3H8,CH4, CO is 20. Calculate the volume of CO2 produced by complete combustion under similar condition 1. 90 ml 2. 160ml 3. 140ml 4. 200ml

SOLVED: Percent by volume is defined as the volume of solute present in 100 mL of solution (%V/V). The formula for percent by volume is: percent by volume = (volume of solute /

PERCENT BY MASS.docx - PERCENT BY MASS/VOLUME WORKSHEET Solution = solute solvent 1 kg = 1000 g 1 L = 1000 m Percent-by-Mass concentration of a | Course Hero

1. What is the formula of expressing concentration by percent by volume? __ ___ 2. Which beaker contains the - Brainly.ph
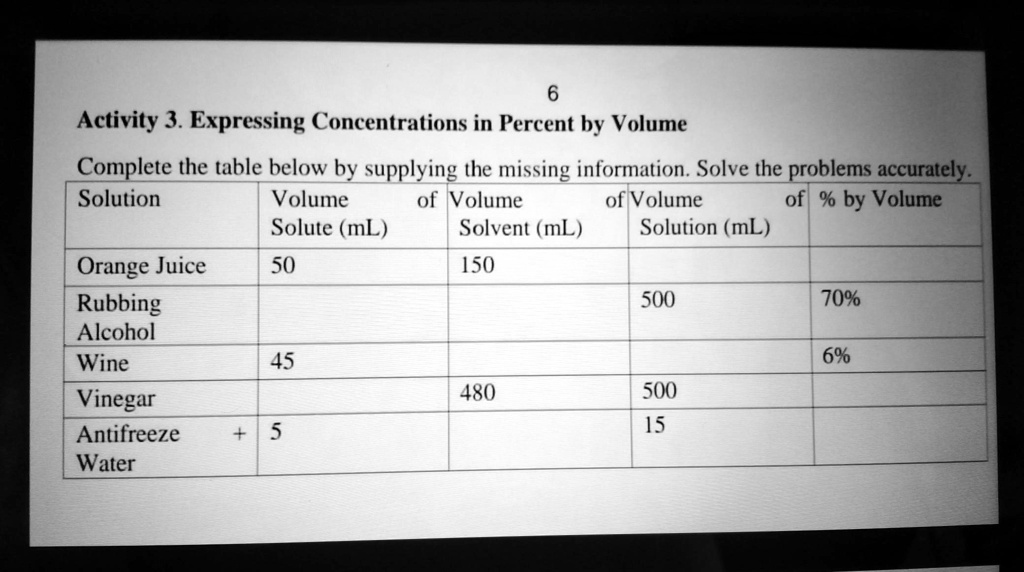
SOLVED: 'Please pa answer thank you! Activity 3. Expressing Concentrations in Percent by Volume Complete the table below by supplying the missing information. Solve the problems accurately: Solution Volume of Volume of












:max_bytes(150000):strip_icc()/GettyImages-493151728-c08e9b2d60bb401b8bbdd2e66de7a93b.jpg)