Indications & Dosing | DARZALEX® (daratumumab) & DARZALEX FASPRO® (daratumumab and hyaluronidase-fihj) HCP
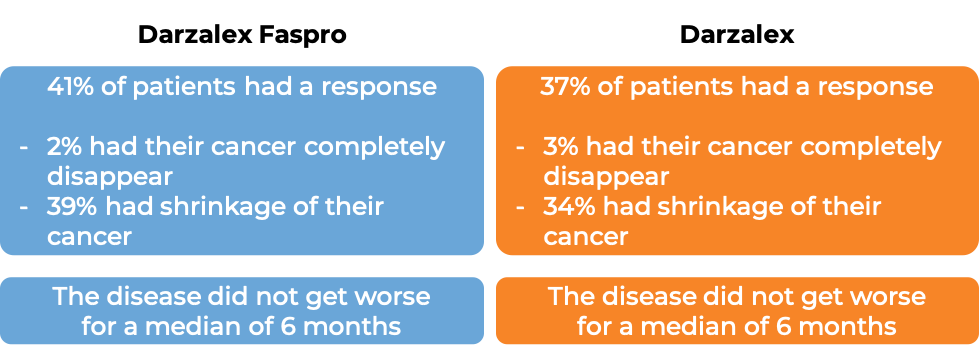
Final analysis of the phase III non-inferiority COLUMBA study of subcutaneous versus intravenous daratumumab in patients with relapsed or refractory multiple myeloma | Haematologica
U.S. Food and Drug Administration Approves DARZALEX FASPRO™ (daratumumab and hyaluronidase-fihj), a New Subcutaneous Formulation of Daratumumab in the Treatment of Patients with Multiple Myeloma

Samer Al Hadidi, MD,MS,FACP on X: "#ASH21 #mmsm Oral sessions on trials @ASH_hematology Talquetamab + Daratumumab ➡️around half pts had previous BCMA therapy ➡️F/U only ~4 mo ➡️ ORR of up to

Daratumumab, carfilzomib, and dexamethasone in relapsed or refractory myeloma: final analysis of PLEIADES and EQUULEUS | Blood Cancer Journal

Indications & Dosing | DARZALEX® (daratumumab) & DARZALEX FASPRO® (daratumumab and hyaluronidase-fihj) HCP
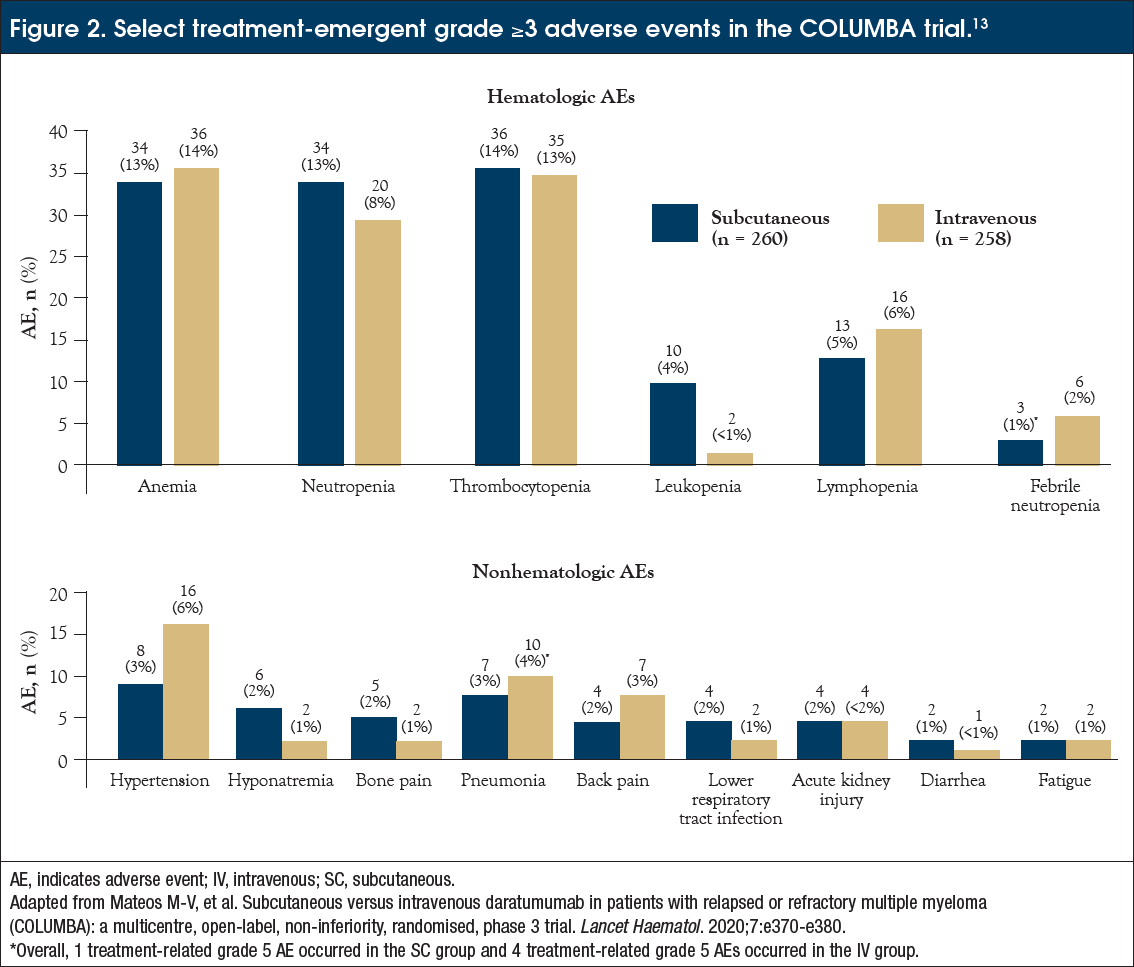
Subcutaneous Daratumumab for Multiple Myeloma: Applying the Lessons from Clinical Trials to Clinical Practice - Journal of Oncology Navigation & Survivorship

Subcutaneous delivery of daratumumab in Japanese patients with relapsed/refractory multiple myeloma | International Journal of Hematology
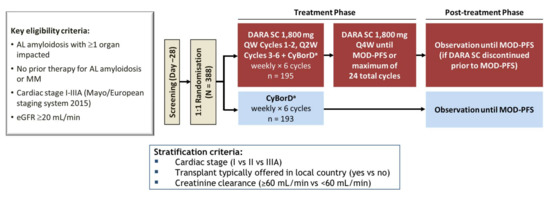
Dosing and administration schema for subcutaneous daratumumab. C1D1,... | Download Scientific Diagram